Not all stem cell treatments are created equal, and the clinic you choose has a direct impact on your safety and outcome. The truth is, the potential side effects of umbilical cord stem cell therapy are not a fixed list; they depend heavily on the quality of the cellular product and the expertise of your medical team. An unapproved product from an overseas clinic carries a vastly different risk profile than a treatment from a compliant U.S. provider. This article will explain how factors like administration methods and FDA compliance influence your safety, ensuring you know how to select a clinic that prioritizes your well-being above all else.
Key Takeaways
- Ask critical questions about safety and compliance: A reputable clinic will be transparent about its FDA-compliant protocols and the source of its cellular products. Verifying these details is the most important step in protecting your health and avoiding unapproved, risky treatments.
- Recognize that mild side effects are often a positive sign: Temporary symptoms like a low-grade fever or soreness at the injection site are typically normal signs that your body’s healing process is starting. Serious complications are rare and usually linked to unregulated clinics or improper administration.
- Ensure your therapy is tailored to your specific needs: Your unique health profile determines if you are a good candidate and which administration method is safest for you. A one-size-fits-all approach is a red flag; effective treatment requires a personalized plan.
What Is Umbilical Cord Stem Cell Therapy?
When you hear about stem cell therapy, you might picture complex, futuristic procedures. But the concept is actually quite simple and grounded in the body’s natural ability to heal. Umbilical cord stem cell therapy uses powerful, young cells sourced from the umbilical cord and placenta, which are collected safely and ethically after a healthy birth. These tissues, once discarded as medical waste, are now recognized as a rich source of mesenchymal stem cells (MSCs) and other regenerative materials.
Unlike other types of stem cell therapy, this method is non-invasive for the donor. The cells are carefully screened, processed, and preserved in a lab, ensuring they are safe and ready for therapeutic use. These cells are prized for their unique properties: they can help manage inflammation, signal your own cells to begin repairs, and promote tissue regeneration. Think of them as a highly effective management team that comes in to direct your body’s own healing crew, helping to address the root cause of pain or degeneration rather than just masking the symptoms.
How Umbilical Cord Stem Cells Work
The magic of umbilical cord stem cells lies in their versatility and potent signaling capabilities. These cells are “undifferentiated,” which means they haven’t yet become a specific type of cell, like a skin cell or a muscle cell. This gives them a remarkable ability to adapt to different environments in the body. When introduced to an area of injury or inflammation, they can release growth factors and proteins that reduce inflammation and encourage damaged tissues to repair themselves.
According to the National Marrow Donor Program, umbilical cord blood is a rich source of hematopoietic stem cells, which can develop into various types of blood cells. While these are well-known for treating blood disorders, the mesenchymal stem cells also found in cord tissue are the key players in regenerative medicine for orthopedic, autoimmune, and anti-aging applications. They act as the body’s natural repair kit, helping to restore function and bring relief.
Conditions Treated with This Therapy
The FDA has approved umbilical cord blood stem cells to treat over 80 different conditions, primarily blood disorders like leukemias, lymphomas, and sickle cell anemia. This established track record highlights their safety and effectiveness in regenerating healthy cells and tissues. You can find a list of FDA-approved stem cell treatments that showcases the extensive history of their use in mainstream medicine.
Beyond these approved uses, the field of regenerative medicine is rapidly exploring how these cells can help with a wider range of issues. At clinics like Miami Stem Cell, we focus on using these therapies for conditions like chronic joint pain from arthritis, sports injuries, back pain, and autoimmune disorders. The goal is to use the anti-inflammatory and regenerative power of these cells to help the body heal itself, offering a promising alternative to surgery or long-term medication.
Types of Umbilical Cord Products
Not all umbilical cord therapies are the same. The products used can be derived from different parts of the umbilical cord, primarily the cord blood or the cord tissue itself (known as Wharton’s jelly). Cord blood is rich in hematopoietic stem cells, while the tissue is a dense source of mesenchymal stem cells (MSCs), which are particularly useful for repairing cartilage, muscle, and other tissues.
These products are administered as umbilical cord stem cell injections, which have shown great results in treating joint pain and soft tissue injuries. The specific product your doctor recommends will depend on your condition. At Miami Stem Cell, we use products that contain a powerful mix of live, healthy MSCs, growth factors, cytokines, and exosomes to provide a comprehensive approach to healing and regeneration.
What Are the Common Side Effects?
One of the first questions people ask about any new treatment is, “What are the side effects?” It’s a great question, and when it comes to umbilical cord stem cell therapy, the answer is reassuring. The vast majority of side effects are mild, temporary, and a normal part of your body’s healing response. Think of it as your system getting acquainted with the powerful new cells that have arrived to help with repairs. Most patients find these effects are minor and resolve quickly, allowing them to get back to their lives with minimal disruption.
Understanding Mild Systemic Responses and Fever
After your treatment, you might feel a bit tired or notice a slight, low-grade fever. This is what’s known as a mild systemic response. It’s a sign that your immune system is interacting with the new stem cells and kicking into gear. This response is generally short-lived and is a positive indicator that the therapy is beginning to work. While it might feel like you’re coming down with a minor cold, it’s simply your body’s natural reaction to the introduction of powerful regenerative cells. We provide detailed information on what to expect after our stem cell treatments so you feel prepared and confident.
Reactions at the Injection Site
Just like with any injection, it’s common to experience some temporary discomfort right at the treatment site. You might notice a little redness, swelling, or tenderness for a day or two. Some people also report temporary stiffness in the area. These localized reactions are completely normal and are typically very mild. They are simply your body’s immediate response to the injection process itself, not a cause for concern. These symptoms usually fade quickly on their own as the area heals and the stem cells begin their work. You can find answers to more procedural questions on our FAQs page.
How to Manage Temporary Symptoms
The good news is that these common side effects don’t last long. Most resolve on their own within a few days, often within 8 to 72 hours. The best thing you can do is listen to your body. Get plenty of rest, stay hydrated, and avoid strenuous activity for the first day or two. We will give you specific post-procedure instructions tailored to your treatment. Following these guidelines is the best way to ensure a smooth recovery. Of course, if you ever have any concerns or questions after your procedure, we encourage you to contact us immediately. We’re here to support you every step of the way.
What Are the Serious Risks and Complications?
While many people experience only minor, temporary side effects, it’s important to be aware of the more serious complications that can arise, particularly with unapproved or improperly administered treatments. Understanding these risks is the first step in making a safe and informed decision about your health. When procedures don’t follow strict safety and screening protocols, or when they use cells that are not properly sourced, the potential for significant health issues increases. Let’s walk through the main areas of concern so you know exactly what to look for when choosing a provider.
The Risk of Graft-versus-Host Disease (GVHD)
Graft-versus-Host Disease, or GVHD, is a serious condition that can occur when your body receives cells from a donor. Think of it as the opposite of organ rejection. Instead of your body attacking the new cells, the new donor cells attack your body. This immune response can range from a mild skin rash to severe, life-threatening damage to your liver, stomach, or other organs. Symptoms can appear soon after treatment or even months later, sometimes causing chronic issues like lasting skin problems or hair loss. This is a primary reason why using properly sourced and processed umbilical cord products is critical, as it helps avoid the complications associated with using unmatched donor cells.
Severe Infections and Immune System Complications
Any time you introduce something new into the body, there is a risk of infection. With certain types of stem cell therapies, especially those that might affect your immune response, this risk can be more pronounced. Patients can become vulnerable to serious bacterial and viral infections if the products are contaminated or if the procedure isn’t performed in a sterile environment. These infections can be difficult to treat and may lead to significant health problems. At Miami Stem Cell, we use umbilical cord-derived mesenchymal stem cells that are rigorously screened for infectious agents, and all our procedures are performed under strict sterile conditions to minimize this risk.
Long-Term Safety and Tumor Formation Risks
The FDA has issued warnings about unapproved regenerative medicine products for a reason. When stem cells are not sourced, processed, or administered correctly, there can be serious long-term consequences. One of the most significant concerns is the potential for tumor formation. If the cells are manipulated improperly or are not the right type, they could grow uncontrollably after being introduced into your body. This is why it’s essential to choose a clinic that adheres to science-backed protocols and uses high-quality, well-characterized cellular products. You can find more information about our commitment to safety in our frequently asked questions.
How Does the Administration Method Affect Side Effects?
When you’re exploring umbilical cord stem cell therapy, it’s easy to focus on the cells themselves. But how those cells are delivered to your body is just as critical. The administration method plays a huge role in the treatment’s effectiveness and the side effects you might experience. Think of it as the difference between mailing a letter to a specific address versus dropping flyers from a plane; one is targeted and precise, while the other is widespread and less controlled.
Different health concerns require different approaches. A localized joint issue, for example, needs a different delivery method than a systemic condition that affects the whole body. Each technique, from intravenous (IV) infusions to direct injections, comes with its own set of considerations. Understanding these differences is key to making an informed decision about your care and ensuring your treatment plan is tailored to your specific needs and safety. At Miami Stem Cell, we carefully select the administration method that offers the greatest potential for positive results while minimizing risks. We believe in a personalized approach, because the right delivery method ensures the cells reach their target and can begin the healing process effectively.
Risks Associated with IV Delivery
Intravenous (IV) delivery of stem cells is a method used by some clinics, but it comes with significant risks you should know about. The main concern is the quality and viability of the cells being used. Research has shown that many umbilical cord products administered via IV contain few, if any, living stem cells. When non-viable or dead cells are introduced into your bloodstream, they can’t perform their regenerative function and may instead trigger an unwanted immune response without offering any therapeutic benefit. This is why we focus on treatment protocols that ensure cell viability and targeted delivery.
Complications from Intrathecal Injections
An intrathecal injection delivers stem cells directly into the spinal fluid. This is a highly specialized procedure used for certain neurological conditions. Because it involves the central nervous system, it carries a higher risk of complications. One study found that more than half of patients who received intrathecal infusions experienced side effects like fever or headaches. While this method can be appropriate in very specific cases, it requires incredible precision and expertise. We believe in a safety-first approach, which means carefully weighing the risks and benefits before considering such an invasive procedure for any patient.
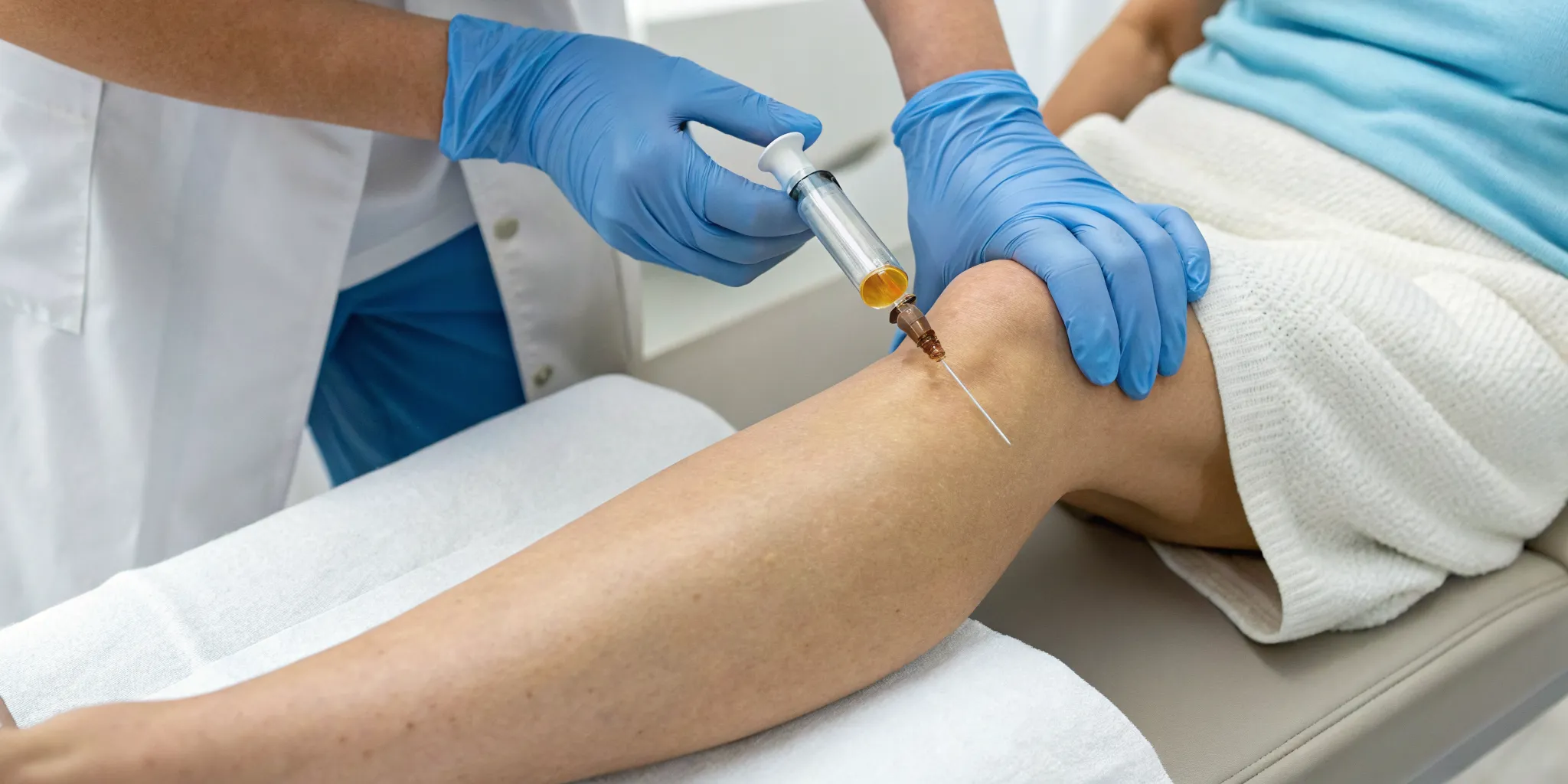
Side Effects from Local Injections
For many conditions, a local injection is the most direct and effective way to administer stem cell therapy. This involves injecting the cells precisely into the area that needs treatment, such as an arthritic knee or an injured shoulder. The side effects from this method are typically mild and temporary. You might experience some localized pain, stiffness, or swelling at the injection site for a few days. This targeted approach allows the cells to get right to work on joint regeneration or tissue repair, minimizing systemic exposure and reducing the likelihood of broader side effects.
What Are the Safety Concerns with Unapproved Treatments?
The field of regenerative medicine is full of promise, but it’s also a space where you need to be careful. Not all treatments are created equal, and the rise of unapproved therapies presents real risks to patients. When clinics operate without proper oversight, they can expose you to ineffective products, unsafe procedures, and serious health complications. Understanding these dangers is the first step toward protecting yourself and making a truly informed decision about your health.
The main concerns fall into a few key areas. First, there’s a lot of confusion around what it means for a treatment to be regulated by the FDA. Many clinics use misleading language that makes their products sound safer than they are. Second, the source of the stem cells and the clinic’s standards matter immensely. Unregulated clinics, especially those overseas, may not follow the strict safety protocols required in the United States. Finally, the quality of the product itself is critical. An effective therapy depends on using viable, healthy cells, which isn’t always what you get with unapproved products.
FDA Approval vs. Registration: What’s the Difference?
It’s easy to get confused by FDA terminology, but the difference between “approved” and “registered” is huge. When a facility is FDA-registered, it simply means they’ve put their name on a list with the government. It does not mean the FDA has reviewed their products for safety or effectiveness. In contrast, FDA approval is a rigorous process where a product is thoroughly tested and scientifically validated to ensure it is safe and works as intended.
Many clinics sell unapproved regenerative medicine therapies by taking advantage of this confusion. They might claim their products are “FDA-compliant” or “FDA-registered,” leading you to believe the treatment has been given a green light. In reality, these products haven’t been proven to be safe or effective, leaving you to take all the risk.
The Dangers of Unregulated and Offshore Clinics
Choosing a clinic without proper credentials or one that operates outside of U.S. regulatory standards can be a dangerous gamble. Health officials have issued warnings about unapproved stem cell products because many are not only unproven but can also be harmful. These clinics may cut corners on safety protocols, leading to risks like contamination, incorrect dosing, or the transmission of infectious diseases.
Clinics located overseas often market themselves as offering cutting-edge treatments unavailable in the U.S., but this is usually because they aren’t held to the same safety standards. When you receive overseas vs. USA stem cell treatments, you have little to no legal recourse if something goes wrong. Sticking with a reputable, compliant clinic in the U.S. ensures your treatment is performed under strict guidelines designed to protect your health.
Why Product Quality Matters: Dead vs. Living Cells
For a stem cell treatment to have any chance of working, the cells must be alive, healthy, and functional when they are administered. Unfortunately, many clinics use off-the-shelf umbilical cord products that contain very few, if any, living cells. These products are often processed, frozen, and shipped, and the cells don’t survive the journey. Some studies have shown that these products contain mostly dead cellular debris.
Clinics making bold claims about their products containing “millions of live stem cells” are often being misleading. Without living, viable stem cells, the treatment has no biological mechanism to promote healing. This is why the source of the cells and the processing methods are so important. A reputable clinic will be transparent about its cell sourcing and processing to ensure you receive a high-quality product with the potential to be effective.
Who Should Avoid Umbilical Cord Stem Cell Therapy?
While umbilical cord stem cell therapy offers incredible potential for healing and regeneration, it’s not the right fit for everyone. Your safety and the treatment’s effectiveness depend on a careful evaluation of your unique health profile. Certain conditions and circumstances can increase risks or reduce the likelihood of a positive outcome. That’s why a thorough screening process is the first and most important step in any regenerative medicine plan. Understanding who might not be an ideal candidate helps you make a fully informed decision about your health.
Who Isn’t a Good Candidate for Treatment?
Certain health conditions can make umbilical cord stem cell therapy unsuitable. A primary concern is the risk of an adverse immune reaction. When using cells from a donor, there’s a possibility of Graft-versus-Host Disease (GVHD), a serious condition where the donor’s immune cells attack the recipient’s body. While the mesenchymal stem cells we use have a low risk of causing this, it’s a factor we carefully consider. Individuals with active cancer, certain blood disorders, or a known allergy to dimethyl sulfoxide (a cryopreservant) are generally not candidates. Our personalized approach ensures we review your complete medical history to determine if this therapy is a safe and viable option for you.
Considerations for Pregnancy and Weakened Immune Systems
For safety reasons, we do not recommend umbilical cord stem cell therapy for women who are pregnant or breastfeeding. There isn’t enough research on how these treatments might affect a developing fetus or a nursing infant, so we always err on the side of caution. Additionally, individuals with severely compromised immune systems may not be good candidates. Because the immune system plays a key role in the healing process, a weakened system can increase the risk of infection. This includes people with active, systemic infections or those taking high doses of immunosuppressive medications. A strong immune response is vital for achieving the best results from any regenerative therapy.
Are There Age-Related Safety Concerns?
Age itself isn’t usually a strict barrier to treatment, but it is an important factor in our evaluation. Research has suggested that very young children may have a higher likelihood of experiencing side effects, as their immune systems are still developing. For older adults, the primary consideration is their overall health rather than their chronological age. Many older patients are excellent candidates and respond well to therapy. However, we carefully assess factors like co-existing chronic conditions and overall frailty to ensure the body is prepared for the regenerative process. The decision is always based on a comprehensive health assessment, not just a number. You can find answers to more common questions on our FAQs page.
What Precautions Should You Take Before Treatment?
Deciding to pursue stem cell therapy is a big step, and it’s smart to be thorough. Taking a few key precautions before your treatment can make all the difference in your safety and results. Think of this as your personal checklist to ensure you’re working with a quality provider and setting yourself up for a positive experience. It’s all about asking the right questions and knowing what to look for. By being proactive, you can feel confident and secure in your decision to explore regenerative medicine for your health goals.
Get the Right Medical Assessments
First things first, a comprehensive medical evaluation is non-negotiable. This isn’t a one-size-fits-all treatment, and any reputable clinic will start with a deep dive into your health history. This process should include a physical exam, a review of your medical records, and may involve specific diagnostic tests to understand the root cause of your condition. This assessment ensures that you are a good candidate for the therapy and allows your medical team to create a personalized treatment plan designed just for you. If a clinic is ready to treat you without a thorough workup, consider it a major red flag.
Verify Your Provider’s Credentials and FDA Compliance
Not all stem cell clinics operate at the same standard. It’s crucial to verify that your provider adheres to strict safety and regulatory guidelines. The FDA has warned about serious harm from unapproved products, so you’ll want to choose a clinic that prioritizes compliance. Ask direct questions: Where do the umbilical cord products come from? What are the lab’s credentials? What safety protocols are in place? A trustworthy provider will be transparent with this information. Choosing a US-based clinic that follows these regulations is a much safer route than seeking treatment overseas, where oversight can be minimal.
Set Realistic Expectations for Your Treatment
It’s easy to get caught up in the excitement of regenerative medicine, but it’s important to have realistic expectations. Be wary of clinics that make grand promises or guarantee a cure. The truth is, many unproven claims are circulating, especially regarding the number of live, functional cells in a product. A responsible doctor will have an honest conversation with you about the potential outcomes for your specific situation. They will explain that healing is a process and that results can vary. A great way to get a feel for real-world results is by reviewing patient testimonials from the clinic you’re considering.
How to Recognize and Manage Side Effects During Recovery
Your recovery is an essential part of your regenerative medicine journey. Knowing what to expect and how to respond to your body’s signals can make the process smoother and safer. While serious complications are rare with treatments that follow strict safety protocols, being informed helps you stay in control of your health. This means understanding the difference between a normal healing response and a sign that you need to call your doctor.
Open communication with your medical team is key. Before your procedure, you should receive clear instructions on what to do during your recovery, including who to contact if you have questions or concerns. Following these guidelines and attending all your follow-up appointments will help ensure you get the best possible results from your therapy.
Warning Signs That Require Immediate Medical Attention
While our treatments are designed for safety, it’s critical to know which symptoms require immediate attention. A rare but serious complication that can occur with unmatched cells is Graft-versus-Host Disease (GVHD). This is an immune reaction that can cause symptoms ranging from a skin rash or mouth sores to more severe issues affecting internal organs. If you notice any of these signs after your treatment, it’s important to seek medical help right away.
Choosing a clinic that adheres to the highest safety standards significantly reduces these risks. The differences between overseas vs. USA stem cell treatments often come down to regulatory oversight and quality control. At Miami Stem Cell, we use carefully sourced umbilical cord-derived mesenchymal stem cells to ensure compatibility and minimize the risk of adverse reactions like GVHD.
What to Expect During Your Recovery
Most patients experience only mild and temporary side effects as their bodies begin the healing process. It’s fairly common to feel a bit of fatigue or run a low-grade fever in the day or two following your procedure. Some studies show that these mild reactions, including fever and sometimes vomiting, happen in about half of patients and typically resolve on their own within 8 to 72 hours.
Think of these symptoms as signs that your body is responding to the therapy. You can usually manage them with rest and hydration. If you received a local injection, you might also notice some temporary soreness, swelling, or redness at the injection site. We provide detailed post-procedure instructions to help you manage these minor effects comfortably at home. You can also find answers to common questions on our FAQs page.
The Importance of Follow-Up Care
Your treatment doesn’t end when you walk out of our clinic. Follow-up care is a crucial step in monitoring your progress and ensuring your long-term safety and success. These appointments allow us to track your healing, manage any lingering side effects, and make sure you are responding well to the therapy. Consistent follow-up helps us confirm that your treatment is delivering the intended benefits.
This ongoing partnership is part of our commitment to your health. We believe in a personalized approach that includes careful monitoring and support throughout your recovery. If you ever have questions or feel that something isn’t right, we encourage you to contact us immediately. Your well-being is our top priority, and maintaining an open line of communication is essential for a successful outcome.
How Miami Stem Cell Prioritizes Your Safety
When you’re exploring regenerative medicine, your safety should always come first. At Miami Stem Cell, we’ve built our entire practice around this principle. We believe that a successful outcome starts with a foundation of trust, transparency, and rigorous safety standards. From your first consultation to your post-procedure care, we take every step necessary to ensure your treatment is not only effective but also administered with the highest level of medical integrity. Here’s a closer look at how we protect your health and well-being.
Our Commitment to FDA-Compliant Protocols
The world of regenerative medicine can be confusing, and unfortunately, some clinics operate without proper oversight. The FDA has warned consumers about unapproved products that haven’t been proven safe or effective. We take this very seriously. At Miami Stem Cell, we strictly adhere to FDA regulations for all our therapies. This means we only use high-quality, ethically sourced umbilical cord-derived mesenchymal stem cells that are minimally manipulated. Our commitment to these protocols ensures you receive a treatment that meets the highest safety and quality standards in the United States, which is a key difference when you compare overseas vs. USA stem cell treatments.
Our Personalized Approach to Screening and Treatment
We believe that effective care is personalized care. Before recommending any treatment, our medical team conducts a comprehensive evaluation of your health history and current condition. This isn’t a one-size-fits-all approach. A thorough screening process is essential to confirm you are a good candidate for therapy and to minimize potential risks, such as adverse immune reactions. By understanding your unique physiology and health goals, we can design a treatment plan tailored specifically to you. This careful, individualized process ensures that our stem cell therapies are not only safe but also optimized for your best possible outcome.
How We Support You After Your Procedure
Our care for you doesn’t end when your procedure is over. We are dedicated to supporting you throughout your recovery. While significant complications are rare, some patients may experience mild, temporary side effects like low-grade fever or soreness at the injection site, which typically resolve within a day or two. We provide you with clear post-procedure instructions and are always available to answer your questions. Follow-up appointments are a crucial part of our process, allowing us to monitor your progress and ensure you are healing correctly. You can read about the experiences of our other patients in our testimonials to see how we stand by them every step of the way.
How to Make an Informed Decision
Choosing to pursue regenerative medicine is a significant step toward taking control of your health. Like any medical procedure, it comes with its own set of considerations. The key is to approach your decision with clear eyes, armed with the right information. This allows you to partner with a provider who prioritizes your safety and aligns with your wellness goals. By understanding both the potential and the precautions, you can feel confident in the path you choose for your care.
Weigh the Risks Against the Potential Benefits
The world of regenerative medicine is exciting, but it’s also filled with clinics making bold claims. The FDA has noted that many unapproved products are advertised to treat various diseases without proven safety or efficacy. It’s important to know that the only stem cell products currently approved by the FDA are specific blood-forming stem cells from umbilical cord blood, intended for certain blood disorders. For other applications, the potential for healing and recovery must be carefully weighed against the risks of unproven treatments. Health officials warn that many stem cell products can be harmful, so a thorough discussion with a qualified physician about the risks and benefits is your most important first step.
Steps to Minimize Complications and Maximize Safety
You can take concrete steps to protect your health and ensure you receive high-quality care. First, verify your provider’s credentials and their commitment to safety protocols. Ask them directly about the source of their products and how they adhere to FDA guidelines. Reputable US-based clinics will be transparent about their processes. You should also research the specific treatment being offered. For example, using unmatched cells from another person for a non-life-threatening condition like knee pain carries unnecessary risks. A trustworthy provider will conduct a thorough medical assessment to confirm you are a good candidate and will only recommend therapies that are appropriate for your specific health needs.
Related Articles
- Umbilical Cord Stem Cell Therapy: Benefits, Safety & What to Expect | Miami Stem Cell
- Stem Cell Therapy Pain for Children with Autism: Safety & Comfort | Miami Stem Cell
- Umbilical Cord Stem Cell Therapy: Benefits, Safety & Guide | Miami Stem Cell
Frequently Asked Questions
Where do the umbilical cord stem cells come from, and is the process ethical? The cells we use are sourced from the umbilical cord and placenta of healthy, full-term births. These tissues, which are normally discarded after birth, are donated by consenting mothers who have gone through a rigorous screening process. The collection is completely safe and non-invasive for both the mother and the baby. This ethical process allows us to use these powerful, young cells to help others heal.
How soon can I expect to see results after the treatment? This is a great question, and the answer is that everyone’s healing journey is unique. Some people notice an improvement in their symptoms within a few weeks as the initial anti-inflammatory effects take hold. However, the real regenerative work happens over time. True tissue repair is a gradual process, so the most significant results often develop over the course of several months as your body works with the new cells to heal from within.
What does the actual procedure feel like? Is it painful? The procedure itself is typically very straightforward and quick. For most conditions, it involves a simple injection directly into the targeted area. You might feel a bit of pressure or temporary discomfort during the injection, much like you would with any other shot. We take every measure to ensure you are comfortable, and most patients report that the process is very manageable.
Why is it so important to choose a clinic that follows FDA guidelines? Choosing a clinic that adheres to strict FDA guidelines is the most important step you can take for your safety. These regulations ensure that the cellular products are sourced, screened, and handled according to the highest standards, which minimizes risks like infection or contamination. It also provides assurance that you are receiving a high-quality product with viable cells, rather than an unproven therapy that may be ineffective or even harmful.
Are the results of this therapy permanent? The goal of umbilical cord stem cell therapy is to provide long-term relief by helping your body repair the underlying cause of your condition, rather than just masking symptoms. For many people, this can lead to lasting improvement in pain and function. However, the longevity of the results can depend on several factors, including the specific condition being treated, your overall health, and your lifestyle. The therapy promotes natural healing, but it doesn’t stop the body’s normal aging process.